ASD Closure Surgery Cost In India 2026
Device closure in India in 2026 costs roughly between Rs 1.5 lakh and Rs 3.5 lakh depending on the hospital, the device used and the city. Open heart surgical repair runs higher, typically between Rs 2.5 lakh and Rs 6 lakh or more at private tertiary centres. But the more important number isn’t the cost. It’s whether the anatomy supports device closure at all because that single question changes the entire cost picture.
“Families often come in having already looked up prices online and the number they find rarely matches what their child’s specific situation actually costs because the defect size, the device choice and whether surgery is even avoidable are all variables that a price list can’t account for” says Dr. Prashant Bobhate, Pediatric Cardiologist in Mumbai, India.
What Drives the Cost of ASD Closure in India?
Not one number fits every child. The cost of ASD closure depends on several variables that shift significantly from one case to the next and understanding them is what stops families from being surprised when the estimate arrives.
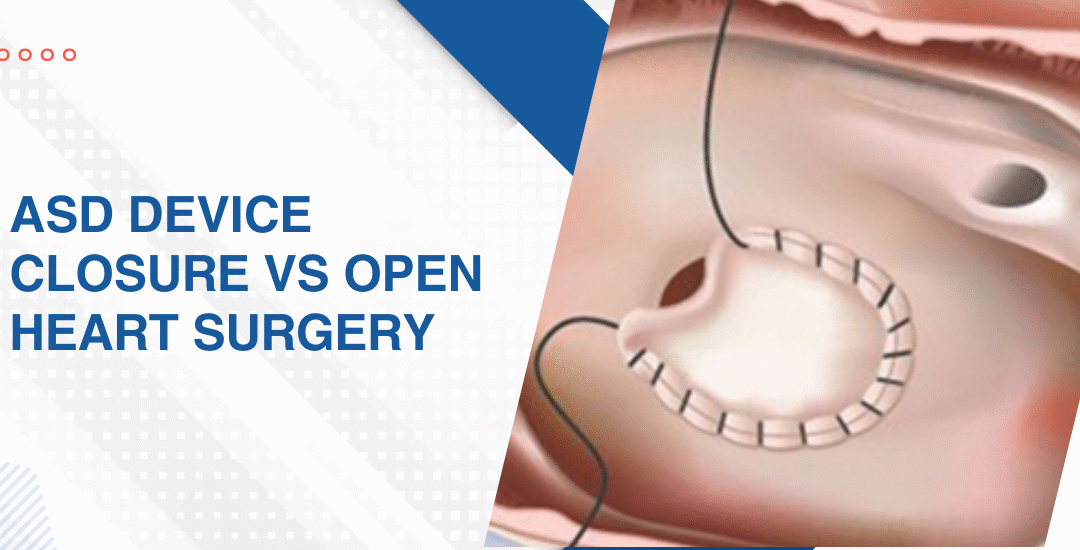
- Device vs surgery: Catheter-based device closure is almost always cheaper than open heart surgery because it avoids the operating theatre, cardiopulmonary bypass, ICU admission and the extended hospital stay that surgical repair requires.
- Device cost itself: The occluder device used in catheter closure is the single largest cost driver in a transcatheter procedure and the price varies depending on brand, size and whether a domestic or imported device is used for that specific defect anatomy.
- Hospital tier: A government or trust hospital will quote significantly less than a premium private tertiary centre for the same procedure and the gap between the two can be substantial without necessarily reflecting a difference in surgical outcome for straightforward cases.
- Defect complexity: A simple mid-septal secundum ASD with good rims costs less to close than a defect with multiple fenestrations, a deficient rim needing a larger device or an anatomy that requires intraoperative TOE guidance throughout a technically demanding catheterisation.
Every child being assessed for ASD closure deserves a detailed structural workup and atrial septal defect evaluation maps the anatomy properly before any cost estimate means anything at all.
What Else Should Families Budget For?
The procedure cost is only part of the picture.
- Pre-procedure workup: Echo, ECG, chest X-ray, blood investigations and in some cases a cardiac MRI or catheterisation for pressure assessment all add to the total before the closure itself happens and families need to factor these in from the start.
- Anaesthesia and OT charges: These are billed separately in most private hospitals and can add Rs 20,000 to Rs 60,000 on top of the procedure estimate depending on the complexity and duration of the catheterisation or surgical session.
- ICU and ward stay: Device closure typically means one to two nights in hospital while surgical repair means a minimum of five to seven days and the daily room and ICU charges in a private tertiary centre accumulate faster than most families expect when they first see the procedure quote.
- Follow-up echos: A device closure requires echo at one month, three months and six months post procedure to confirm device position and residual shunting and these follow-up visits carry their own charges that are rarely included in the initial procedure estimate families receive.
Parents wanting to understand what happens when an ASD goes unrepaired for too long and the pressure consequences that follow should read this piece on when lung transplant becomes necessary for pulmonary hypertension because the cost of delayed correction is always higher than the cost of timely intervention.
Why Choose Dr. Prashant Bobhate for Pediatric Cardiac Care in Mumbai?
Cost transparency matters but so does getting the right procedure recommendation in the first place because a family that pushes for device closure on an anatomy that needs surgery doesn’t save money they create a more complicated and expensive situation down the line. Dr. Prashant Bobhate has spent over 12 years performing catheter-based ASD closures and managing surgical referral pathways for defects where device closure isn’t the right answer at the Children’s Heart Centre, Kokilaben Dhirubhai Ambani Hospital.
Schedule a consultation to find out if a cure is possible and what the right treatment plan looks like for you.
FAQs
Is ASD device closure cheaper than open heart surgery in India?
Yes in most cases because it avoids bypass, ICU and a long hospital stay but the anatomy has to support a device and not every ASD qualifies regardless of what the cost difference looks like.
Does health insurance cover ASD closure surgery in India?
Most major health insurance policies cover ASD closure under congenital heart disease but the extent of coverage, pre-authorisation requirements and whether device cost is included varies significantly between insurers and policy types.
Can ASD closure be done at a government hospital in India?
Yes and the cost at government or trust hospitals is significantly lower than private centres but waiting times, device availability and surgical volumes vary and families need to weigh those factors alongside the price difference.
What happens if ASD closure is delayed due to cost?
An unrepaired ASD continues loading the right heart with excess blood volume and over years causes right heart enlargement, arrhythmia and eventually pulmonary hypertension that makes correction far more complex and expensive than it would have been done early.
References:
- Atrial Septal Defect, MedlinePlus, U.S. National Library of Medicine — https://medlineplus.gov/ency/article/000097.htm
- Congenital Heart Defects, National Heart Lung and Blood Institute — https://www.nhlbi.nih.gov/health/congenital-heart-defects