What Is Rheumatic Heart Disease And How Is It Caused n Children
Rheumatic heart disease is permanent valve damage caused by rheumatic fever, itself triggered by an untreated group A streptococcal throat infection. The immune system meant to fight the bacteria turns on the heart valves instead and that misdirected attack leaves scar tissue that stiffens and narrows valves in ways the heart simply can’t recover from without intervention.
“The tragedy of rheumatic heart disease is that it’s almost entirely preventable and yet I still see children with severely damaged mitral valves from a strep throat written off as a viral fever three or four years earlier,” says Dr. Prashant Bobhate, Pediatric Cardiologist in Mumbai, India.
What Actually Causes Rheumatic Heart Disease in Children?
The bacteria infects the throat and if nobody treats it properly the immune response that follows doesn’t stay where it belongs.
- Strep ignored: An untreated streptococcal throat infection triggers antibodies that cross-react with cardiac tissue because the bacterial proteins look enough like heart valve proteins to confuse the immune system completely.
- Repeated infections: One episode damages the valve somewhat but each recurrent strep infection in a child without penicillin prophylaxis adds another layer of scarring that compounds quietly with every round.
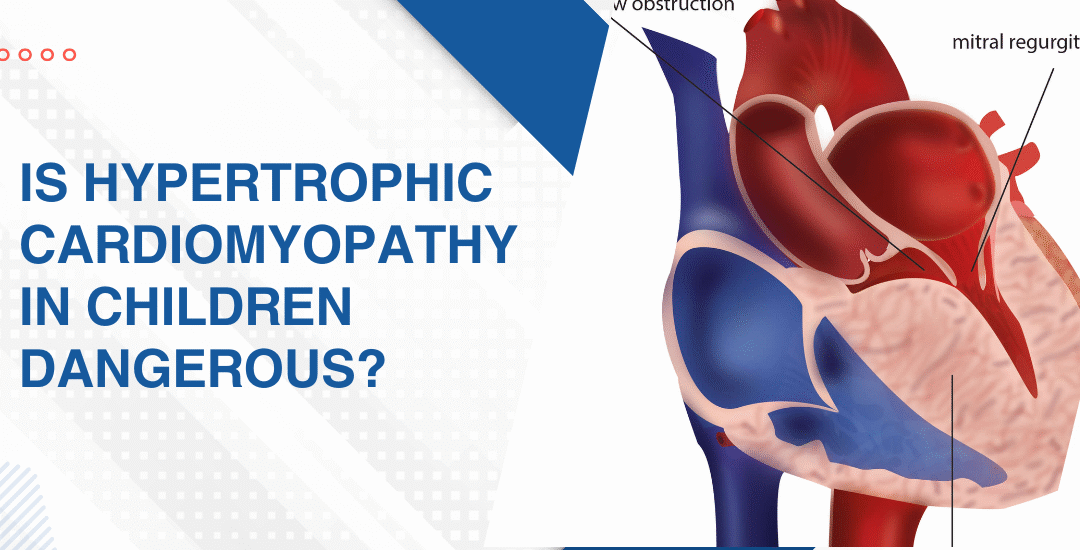
- Mitral valve: This is the valve that takes the worst hit and the leaflets gradually thicken, fuse at the tips and lose the flexibility they need to open and close across a lifetime of beats.
- Poverty drives it: Overcrowded homes, no antibiotics nearby and delayed care all mean strep throat goes untreated long enough to start the whole cascade and India carries a disproportionately high burden of this disease for exactly that reason.
Children showing breathlessness, reduced stamina or a murmur after a history of recurrent throat infections need urgent specialist assessment and pediatric heart failure management starts with knowing exactly what the valves look like right now.
How Is Rheumatic Heart Disease Managed in Children?
Prevention first. Then damage control. Surgery only when the valve has gone too far.
- Penicillin injections: Any child who has had confirmed rheumatic fever needs monthly benzathine penicillin for years, sometimes decades, to stop recurrent strep from adding more damage to what’s already there.
- Echo surveillance: Regular echocardiography tracks how the valve is changing and catches the point where waiting becomes genuinely riskier than stepping in and doing something about it.
- Symptom medications: Diuretics and vasodilators manage the haemodynamic load in symptomatic children who aren’t yet at the threshold where a procedure becomes the better option.
- Balloon or surgery: When mitral stenosis is severe enough to impair output and daily life, balloon dilatation or surgical repair is the only real way to restore forward flow and give the left atrium some relief.
Parents wanting to understand what cardiac warning signs look like in children before any formal valve diagnosis gets made should read this piece on top 5 warning signs of pediatric heart failure because catching it early is always what keeps the most options open.
Why Choose Dr. Prashant Bobhate for Rheumatic Heart Disease Care in Mumbai?
You want someone who can look at the valve on echo, tell you honestly where it’s heading and have the real conversation about what twelve looks like now and what twenty-five looks like if nothing changes. Not a soft answer designed to make the appointment feel better. A plan. Dr. Prashant Bobhate has spent over 12 years managing paediatric valve disease from early subclinical rheumatic damage through severe mitral involvement requiring intervention at the Children’s Heart Centre, Kokilaben Dhirubhai Ambani Hospital.
Schedule a consultation to find out if a cure is possible and what the right treatment plan looks like for you.
FAQs
Can rheumatic heart disease be cured in children?
The damage already done can’t be undone but progression stops with consistent penicillin prophylaxis and severely damaged valves can be repaired or replaced when intervention becomes necessary.
How common is rheumatic heart disease in India?
Very common. Delayed antibiotic access, overcrowding and late diagnosis mean strep throat progresses to rheumatic fever here far more often than it should and India carries one of the highest RHD burdens globally.
What are the first signs of rheumatic heart disease in a child?
Breathlessness during activity, reduced stamina, joint pains after a recent throat infection or a newly detected murmur are the signs that should send a family straight to a paediatric cardiologist without waiting.
Does every child with rheumatic fever develop heart disease?
Not every child but around 30 to 45 percent with untreated rheumatic fever develop some valve damage which is exactly why penicillin prophylaxis after any confirmed episode isn’t optional.
References:
- Rheumatic Heart Disease, MedlinePlus, U.S. National Library of Medicine — https://medlineplus.gov/rheumaticheartdisease.html
- Rheumatic Heart Disease, World Heart Federation — https://world-heart-federation.org/programmes/rheumatic-heart-disease